|
The process of evaporation occurs at a temperature just below the boiling point of the liquid. The temperature at which the liquid becomes a gas is known as the boiling point. If thermal energy continues to be provided, the particles move faster and faster and a change of state occurs where the liquid becomes a gas. This process by which the particles leave the liquid is known as evaporation. The particles are moving at such a speed that they are able to leave the liquid. If enough thermal energy is provided, all of the forces of attraction between the liquid particles are overcome. The faster the particles move, the bigger the gaps between the particles and the weaker the forces of attraction between them. 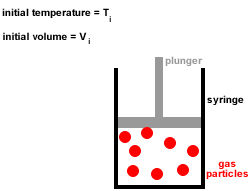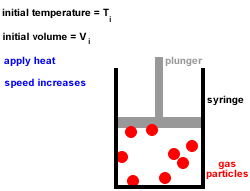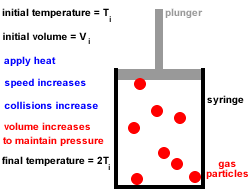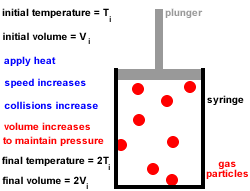
When thermal energy is provided to a liquid, the particles gain kinetic energy and move faster. The temperature at which the liquid becomes a solid is known as the freezing point. If enough heat energy is removed, the liquid turns into a solid. The distance between the particles decreases and the particles become so close that the forces of attraction between them increase in strength. The particles have less kinetic energy and begin to move slower. When a liquid is cooled the thermal energy is transferred from the liquid to the surroundings. The temperature at which a solid becomes a liquid is known as its melting point. 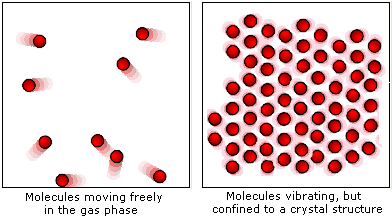
The ice cube melts to liquid water if enough heat energy is provided. For example, if an ice cube is heated, the thermal energy is transferred to kinetic energy and the particles start to move more. This causes the solid to expand and turn into a liquid. The particles move free from their fixed positions and gaps form between them. Once enough heat energy is provided, the vibrations of the particles may be so vigorous that the forces of attraction between them are too weak to hold them in place any longer. The more heat energy is provided, the more kinetic energy is provided and the more the particles vibrate. When a solid is heated thermal energy is transferred to particles as kinetic energy and they vibrate in their fixed positions. The arrangement of particles in solids, liquids and gases is shown in the diagram below: This makes the gaps between the particles larger, causing the gas to expand and the pressure of the gas to increase. When a gas is heated, the particles in a gas gain kinetic energy and move faster. Gases do not have a fixed shape or volume and this enables them to flow and fully fill a container as they travel quickly in different directions. There are very big gaps between the particles in a gas which means that the particles can move easily past each other and gases can flow. This allows the particles to move constantly in a random pattern. The particles in a gas have very weak forces of attraction between them. 
As the particles in liquids can therefore move past each other, liquids do not have a fixed shape and are able to flow. This movement creates gaps between the particles which other particles are able to move into. In a liquid, the particles move continually in a random arrangement, meaning there is no specific pattern of their movement. The particles in a liquid are still close together but the forces between the particles are weaker than those between the particles in a solid. The particles in a solid can only vibrate in their fixed position. 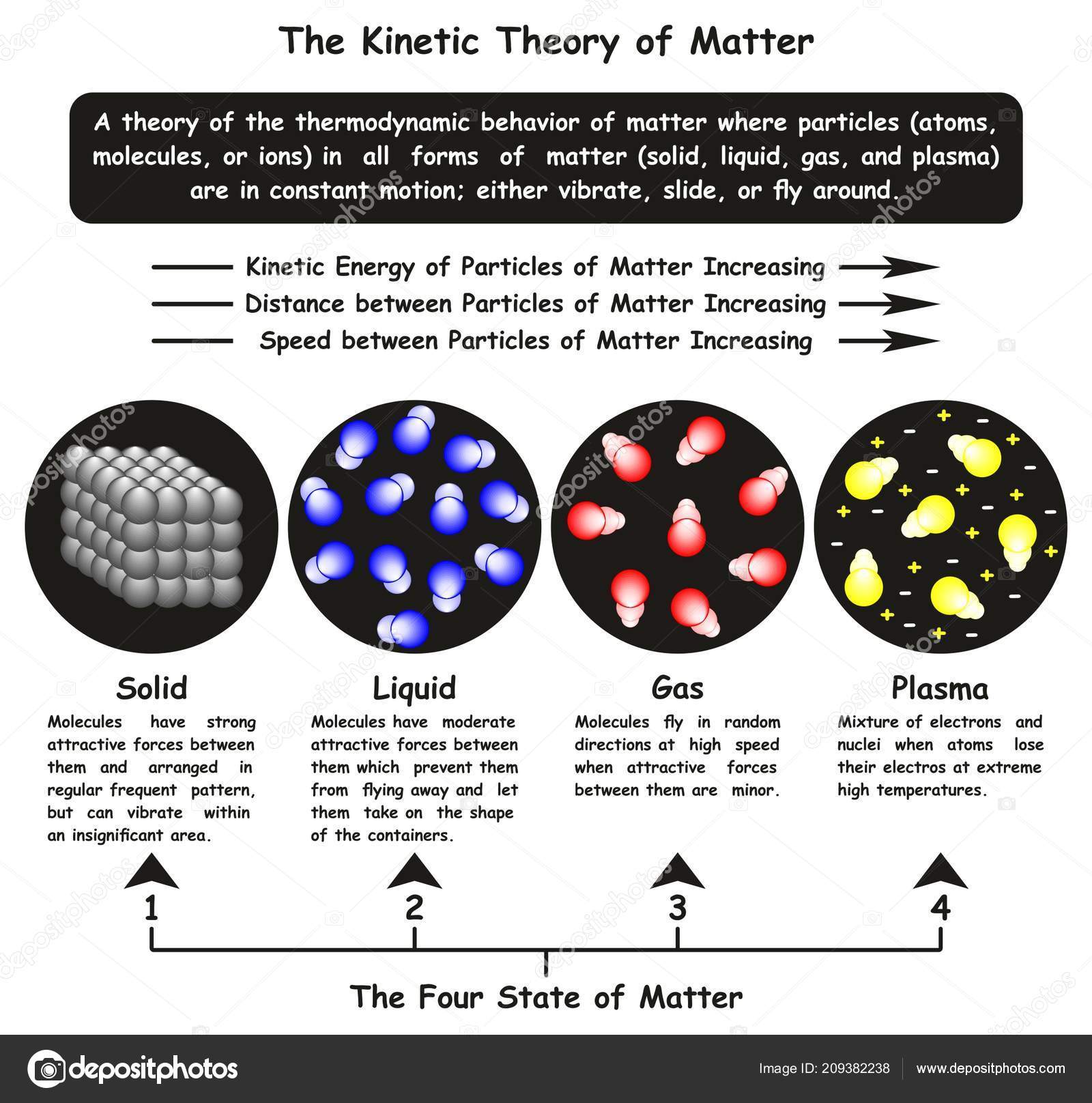
As the particles are fixed tightly together, with no spaces between them, they cannot move or flow, and solids cannot be compressed. This means that solids have fixed shapes as their particles are very close together with a regular arrangement. The particles in solids have strong forces of attraction between them. These different arrangements of particles can be used to explain the differences in their properties and behaviours. The arrangement of particles differs in the three states of matter – solids, liquids and gases. Mean inter-particle distance (or mean inter-particle separation) is the mean distance between microscopic particles (usually atoms or molecules) in a macroscopic body.Everything around us is made up of particles. JSTOR ( January 2012) ( Learn how and when to remove this template message).Unsourced material may be challenged and removed.įind sources: "Mean inter-particle distance" – news Please help improve this article by adding citations to reliable sources. This article needs additional citations for verification.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
